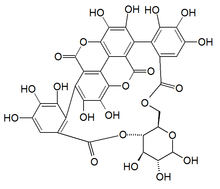
Punicalin
 | |
| Names | |
|---|---|
| Other names 4,6-(S,S)-Gallagyl-D-glucose | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
InChI
| |
| |
| Properties | |
Chemical formula | C34H22O22 |
| Molar mass | 782.52 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Punicalin is an ellagitannin. It can be found in Punica granatum (pomegranate)[1] or in the leaves of Terminalia catappa, a plant used to treat dermatitis and hepatitis.[2][3] It is also reported in Combretum glutinosum, all three species being Myrtales, the two last being Combretaceae.
It is a highly active carbonic anhydrase inhibitor.[4]
Chemistry
The molecule contains a gallagic acid component linked to a glucose.
References
- ^ Zhang, Y.; Wang, D.; Lee, R. P.; Henning, S. M.; Heber, D. (2009). "Absence of Pomegranate Ellagitannins in the Majority of Commercial Pomegranate Extracts: Implications for Standardization and Quality Control". Journal of Agricultural and Food Chemistry. 57 (16): 7395–7400. doi:10.1021/jf9010017. PMID 20349921.
- ^ Lin, C. C.; Hsu, Y. F.; Lin, T. C.; Hsu, F. L.; Hsu, H. Y. (1998). "Antioxidant and hepatoprotective activity of punicalagin and punicalin on carbon tetrachloride-induced liver damage in rats". The Journal of Pharmacy and Pharmacology. 50 (7): 789–794. doi:10.1111/j.2042-7158.1998.tb07141.x. PMID 9720629. S2CID 8639399.
- ^ Lin, Chun-Ching; Hsu, YF; Lin, TC (1999). "Effects of punicalagin and punicalin on carrageenan-induced inflammation in rats". The American Journal of Chinese Medicine. 27 (3 & 4): 371–376. doi:10.1142/S0192415X99000422. PMID 10592846.
- ^ Satomi, H.; Umemura, K.; Ueno, A.; Hatano, T.; Okuda, T.; Noro, T. (1993). "Carbonic anhydrase inhibitors from the pericarps of Punica granatum L". Biological & Pharmaceutical Bulletin. 16 (8): 787–790. doi:10.1248/bpb.16.787. PMID 8220326.
- v
- t
- e
Types of ellagitannins
- Diphenolic acid : Hexahydroxydiphenic acid (HHDP)
- Triphenolic acids : Nonahydroxytriphenic acid
- Sanguisorbic acid
- Valoneic acid
- Tetraphenolic acids : Isoterchebulic acid
- Terchebulinic acid
- Ellagic acid
- Gallagic acid
- Gallagic acid dilactone
- Luteic acid
- Sanguisorbic acid dilactone
- Valoneic acid dilactone
- Acetonyl geraniin
- Alnusiin
- Bicornin
- Carlesiin
- Casuarictin
- Emblicanin A and B
- Euscaphinin
- Galloyl pedunculagin
- Grandinin
- Helioscopinin B
- Jolkinin
- Lagerstannin A, B and C
- Macranganin
- Myrobalanitannin
- Nupharin A, B, C, D, E and F
- Pedunculagin
- Punicalagin
- Punigluconin
- Phyllanemblinin A, B, C, D, E and F
- Punicalin
- Roburin E
- Rugosin E
- Sanguiin H-5
- Stenophyllanin A, B and C
- Strictinin
- Tellimagrandin I and II
- Teracatain
- Terchebulin
- Terflavin A and B
- Tergallic acid
- Tergallic acid dilactone
| C-glycosidic ellagitannins |
| ||||
|---|---|---|---|---|---|
| Dehydroellagitannins (molecules with dehydrohexahydroxydiphenic acid (DHHDP) |
| ||||
| Transformed ellagitannins |
|
- Dimers
- Agrimoniin
- Cornusiin E (dimer of tellimagrandin II)
- Lambertianin A and B
- Nobotanin B
- Roburin A, B, C and D
- Sanguiin H-6
- Trimers
- Lambertianin C
- Raspberry ellagitannin
- Tetramers
- Lambertianin D
- Nobotanin S
- Pentamer
- Melastoflorin A
- Flavono-ellagitannins (hybrid type)
- Urolithins (metabolites)
- Pomegranate ellagitannins
 | This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











